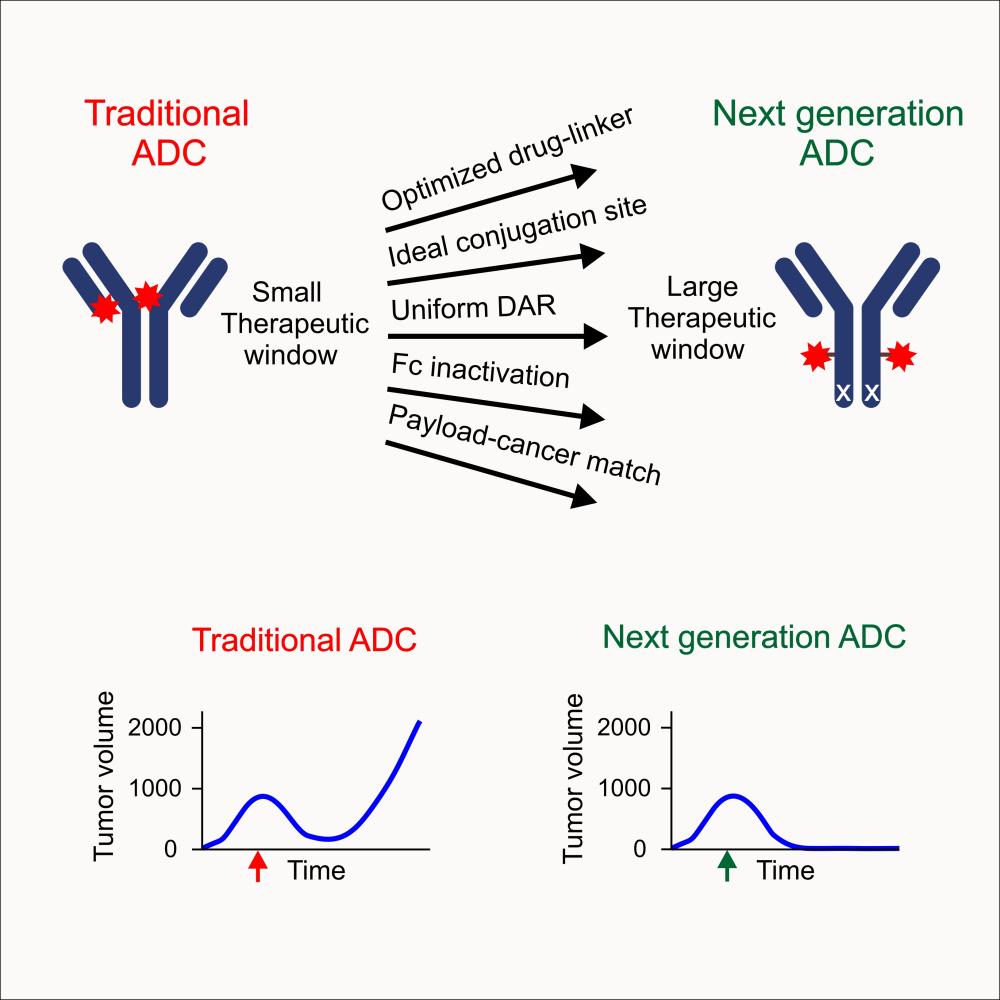
In cancer therapeutics, targeted therapies are a rapidly developing field of interest. These include antibody-drug conjugates (ADCs), compounds that carry small-molecule anticancer drugs to specific antigens on tumors, directly killing tumor cells. Though the first ADC approved by the U.S. Food and Drug Administration (FDA) was brought to the clinic in 2000, it was later voluntarily removed due to high toxicity. Since then, more precise technologies have improved generations of ADCs, and there are currently 12 ADCs that are FDA-approved for clinical use. However, most available ADCs offer limited improvement and can still be toxic.
But a study conducted by researchers in the National Cancer Institute’s Center for Cancer Research and published in Cell Reports in December 2023 highlights a new ADC—carefully engineered, screened, and purified—that can eradicate large tumors in animal models at a dose per body weight that humans can tolerate, showing that ADCs can be improved and made less toxic. Clinical trials with humans will be needed to prove efficacy.
The ADC had potent effects in mouse studies and was capable of eliminating tumors larger than those typically tested in mice, reaching sizes large enough to be identified by self-examination in humans.
“If you really want to create a therapy that’s going to be effective in humans, I think the bar should be higher. You should be able to treat a tumor that’s this size and still see potent activity,” said Brad St. Croix, Ph.D., a senior scientist at NCI Frederick and the senior author on the study.
The team’s ADC proved to be remarkably effective at abolishing tumors this size in breast cancer mouse models—and with a dose much lower than that typically used for ADCs in preclinical studies, meaning the therapeutic index, a measure of effectiveness versus safety, is very high.
The study’s authors, including St. Croix and first author Yang Feng, Ph.D., have been working on this therapy for more than a decade. They set out to create an ADC with limited off-target toxicities, but they encountered some surprising discoveries along the way.
‘The Final Form is Better Than the Sum of Its Parts’
Feng and St. Croix study how the tumor microenvironment—the normal cells and blood vessels that the cancer cells pirate to support themselves—promotes tumor growth, and how to exploit those mechanisms to develop improved therapies.
Antibody therapy is one way to do it, since antibodies can be designed to target specific molecules on the surface of tumor-associated cells. The protein the team targeted for their study, known as CD276, is “one of the best [targets] we’ve ever encountered,” said St. Croix. It’s dramatically overexpressed in the tumor and tumor-associated cells of the tumor microenvironment, but not in normal cells outside of the tumor. In theory, that meant an antibody directed against it would strike fewer healthy cells and be less toxic to the patient.
But just targeting the tumor by hitting CD276 isn’t enough because, the team found, CD276’s functional role isn’t essential for the tumor’s viability. They previously discovered that even if they destroyed its function by eliminating the CD276 gene in cancer cells or tumor-associated microenvironmental cells in mice, the tumors continued to grow, suggesting that loss or lack of CD276 was insufficient for blocking tumor growth.
So, their antibody blocking CD276 needed to be combined with a drug to make it more effective. The team chose a pyrrolobenzodiazepine, a potent DNA-damaging drug. Like a directed missile, the antibody brings the drug to the tumor cells and its associated microenvironment. Because the ADC is so potent, only a little is needed to kill the tumor.
Essentially, we “added a warhead to the naked antibody to create the ADC,” said Feng. “With ADCs, the antibody’s function-blocking ability is not as relevant as its ability to bind to the target. Because the drug payload allows the direct killing of target-expressing cells, with ADCs, the final form is better than the sum of the parts.”
But the scientists knew this potent combination would likely still produce undesirable effects, since off-target toxicities are a common challenge with ADCs. Their goal then became fine-tuning their ADC to make it as useful as possible while causing as little harm as possible, and in ways that might one day be applied to other ADCs.
Fine-Tuning the ADC
The team had some advantages using different techniques than what had previously been tested. Unlike traditional methods, they created the antibodies in the laboratory instead of in mice. This meant that the antibodies were fully human from the get-go and didn’t need to be tweaked further to reduce the odds that they could be recognized as foreign by the recipient’s immune system, which could streamline their path to the clinic. The team also made the antibodies react to CD276 of both humans and mice, so that they’d yield accurate results in laboratory tests.
St. Croix’s group, part of NCI’s Mouse Cancer Genetics Program, also took advantage of their previously developed strain of mice that lack CD276. That let the team measure the ADC’s toxicity in both normal mice and the knockout mice to determine whether the ADC was hitting the tumor or causing wider toxicity by killing healthy cells.
The results surprised them: The toxicity was the same.
The team determined the ADC wasn’t specific enough and revisited its design and purification to improve its ability to target just cells with CD276.
Their work paid off. Upon testing again, they could see more harm in the CD276-expressing mice than the mice without CD276, suggesting the predominant off-target toxicities had diminished. Some of the complex pieces were finally starting to work together.
And, as a bonus—“It seemed like with every step we took to make it less toxic, it seemed to, at the same time, become more potent, which may be due to increased drug availability at the tumor site,” said St. Croix.
A Long Process, with Help Along the Way
Though the pieces fell into place, the road to developing the ADC was long and complicated.
ADCs are complex, and this one required time and the help of other specialists—from medicinal chemists to animal technicians to imaging laboratories and other—to understand and fine-tune each component. “We are a research lab, and we have our focus,” said Feng. “But this ADC design actually takes more specialty than we have. That’s why we have so many collaborators.”
“This is why it’s been very good to do this sort of work here at the NIH … the NIH has many different labs and cores with different complementary skill sets that were necessary for this sort of a study,” said St. Croix.
Looking to the Future, with Hope
Now that the study has concluded, the team is already eyeing next steps.
Their technology was recently licensed by BrickBio Inc., a Boston-based biotechnology company, whose goal is to try and bring the CD276 ADC to the clinic based on the team’s research.
But it may be a long time yet until that happens. Going to clinical trials is an intricate process. Additionally, some steps of the process may need to be calibrated further to make sense during scale-up in drug manufacturing and to further eliminate residual toxicities.
Clinical trials in human patients will be necessary to answer the next round of questions. But the team is hopeful.
“What excites me is the potential to help people,” said St. Croix. “I think [our ADC] has tremendous potential, especially for breast cancer patients.”
Karolina Wilk is a technical editor in Scientific Publications, Graphics & Media (SPGM), where she writes for NCI Frederick and Frederick National Laboratory’s news outlets and edits scientific manuscripts, corporate documentation, and other writing. SPGM is the facilities’ creative services department and hub for editing, illustration, graphic design, formatting, and multimedia training and support.