A study originating in NCI Frederick’s RNA Biology Laboratory has clarified why cells pack up and discard a certain type of RNA.
Put simply, it’s a trash removal system meant to keep cells stable—and it’s relevant to cancer growth.
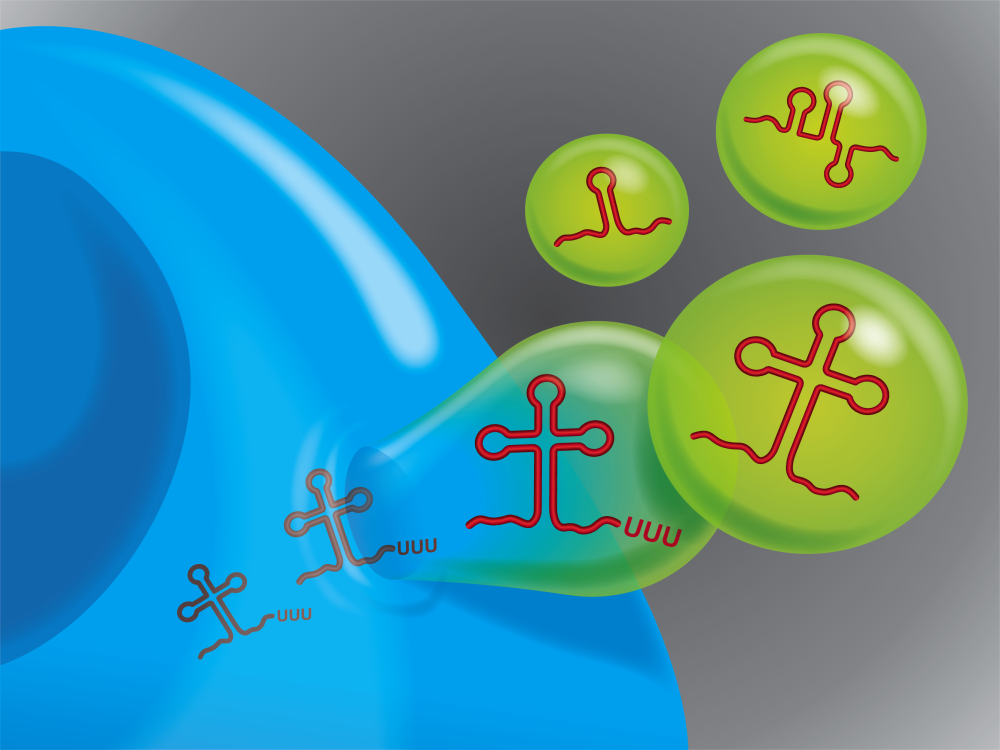
The findings, published in the Proceedings of the National Academy of Sciences, came by examining the RNAs within extracellular vesicles (EVs).
What is RNA?
RNA is a sort of companion to DNA, responsible for assembling proteins, activating certain genes, and many other crucial biological functions. EVs, particles containing cellular components such as RNA and proteins, get released from cells to initiate or complete various tasks. Noncoding RNA is the most prevalent type of RNA in EVs.
What Were the Findings?
Through their examination, the study team demonstrated that the noncoding RNAs in EVs are defective. Many are cast-off, functionless byproducts of other cellular processes. Some of them are deformed or fragmented. Others have molecular additions that change their chemical structures.
Putting these abnormal RNAs into EVs and shipping them out is one way for cells to eliminate them, the team found.
“We learned that extracellular vesicles are part of a new pathway to remove aberrant RNAs from cells,” said Sandra Wolin, M.D., Ph.D., chief of the RNA Biology Laboratory and senior author on the study.
The study was also led by Sandra Williams, M.D., Ph.D., the Metzger scholar in Translational Research at the National Institute of Arthritis and Musculoskeletal and Skin Diseases.
Up to now, scientists knew that noncoding RNA was prevalent within EVs, but they didn’t know why or what purpose its presence served.
The contain-and-discard method backs up other cellular clean-up mechanisms, according to the study. There’s already an enzyme, DIS3L2, that breaks down certain abnormal RNA inside cells. As part of the analysis, the team modified a mouse cell line (a stand-in to understand other mammalian cells, including human cells) to reduce DIS3L2 levels. This led to an increase in defective noncoding RNA in EVs, suggesting the cells were using the EVs to compensate.
The team also blocked the formation of EVs in the cell line to learn what happens when cells can’t break down or discard the defective RNA. The resulting buildup of abnormal RNA activated cells’ internal antiviral defenses as the cells tried to deal with the problem.
The Connection to Cancer
Since abnormal RNAs can contribute to cancer, “there is now evidence that these RNAs can activate these [defensive] sensors to influence the growth of tumors,” Wolin said.
She explained tumor cells with mutations that make the RNA removal mechanisms less effective could trigger a stronger defensive response, which in turn could lead to a greater immune system response against a tumor.
On the other hand, tumor cells can acquire mutations that make their defective RNA removal especially efficient, decreasing their likelihood of being noticed and targeted by the immune system, she said.
“By uncovering novel pathways by which cells recognize and remove excess, defective, and unneeded RNAs, our work could advance understanding of the ways in which RNA surveillance pathways can be manipulated to influence the tumor microenvironment,” Wolin said.
Samuel Lopez leads the editorial team in Scientific Publications, Graphics & Media (SPGM). He writes for newsletters; informally serves as an institutional historian; and edits scientific manuscripts, corporate documents, and sundry other written media. SPGM is the creative services department and hub for editing, illustration, graphic design, formatting, and multimedia.