Early in the COVID-19 pandemic, before vaccines were available and while SARS-CoV-2 was wreaking havoc worldwide, Barry O’Keefe, Ph.D., and longtime collaborators in Brazil and Spain pooled their knowledge and resources to study cyanovirin-N (CV-N), a protein from cyanobacteria, photosynthetic bacteria commonly known as blue-green algae. O’Keefe had a hunch it could be developed to fight the spread of SARS-CoV-2.
The team’s study, published this spring, suggests that it may. They not only determined that CV-N blocks the virus’ ability to infect cultured human cells and animals, they also figured out the molecular mechanism behind how it works—and that it might work against all human coronaviruses.
“In summary, we present an anti-coronavirus agent that works by an unexploited mechanism and prevents infection by a broad range of SARS-CoV-2 strains,” said O’Keefe, director of the Molecular Targets Program (MTP) at NCI Frederick and the chief of NCI’s Natural Products Branch.
CV-N is a lectin, a type of natural protein found in plants, algae, and some microbes. Lectins have been studied extensively as antiviral treatments because many can halt certain viruses by binding to glycoproteins (proteins with sugar molecules attached) on the viral surface envelope.
MTP’s contribution to this collaboration was to “interrogate cyanovirin’s source of activity against SARS-CoV-2” to find out exactly how it might block the virus, said Lauren Krumpe, a protein chemist and molecular biologist in the MTP.
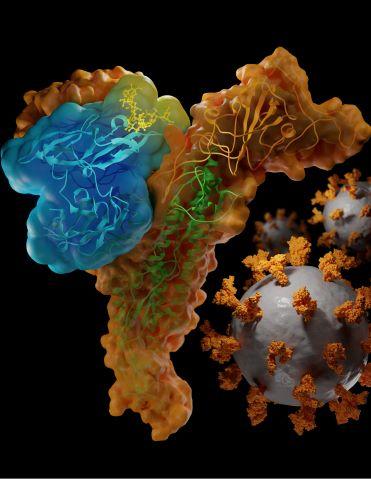
CV-N Keeps Spike Protein from Allowing Viral Invasion
Krumpe performed the analyses to figure out where CV-N binds to SARS-CoV-2. Her results showed that CV-N attaches to the Spike protein, the “hook” the virus uses to grapple and enter a host’s cells to cause an infection. Specifically, CV-N interacted with a region within the Spike protein called the S1 domain.
“Everyone was excited,” Krumpe said of the findings. Binding to Spike meant that CV-N might stop SARS-CoV-2 from infecting human cells.
The next step was to evaluate this interaction on a more detailed level. Using mass spectrometry to analyze small pieces of the S1 domain mixed with CV-N, Krumpe made another intriguing discovery: CV-N doesn’t interact with the receptor-binding domain (RBD) portion of S1, the typical target of most SARS-CoV-2 vaccines and treatments. Rather, it attaches to a few specific oligosaccharides (which are a type of carbohydrate) that adorn a different portion of S1. Subsequent cell culture studies showed that CV-N’s interaction with those molecules keeps the envelope of SARS-CoV-2 from fusing with the host cell.
This was “great news and potentially interesting science,” Krumpe said, because this raised the possibility of using CV-N as a prevention or treatment.
This finding was key because the RBD has become increasingly mutated as SARS-CoV-2 variants have emerged. That’s what has made vaccines less effective at protecting people from infection and reinfection.
So, finding out that “CV-N has very minimal to no interaction with RBD” piqued the group’s interest, said Krumpe. Since CV-N can bind to the Spike protein and keep the virus from infecting cells no matter what mutations the RBD may have, developing it as an anti-coronavirus treatment may offer an effective method for managing future outbreaks.
That suggests CV-N could become a tool to reduce coronavirus transmission and infection when vaccines aren’t yet available or when they aren’t effective at stopping transmission.
“We need vaccines that prevent people from going to the hospital in first place, but we could also use something like these lectins that would prevent the transmission of the virus,” O’Keefe said.
Developing CV-N into a treatment to prevent coronavirus infection would follow in the steps of griffithsin, another lectin that is now in Phase 1 clinical trials to test its safety in humans as a nasal spray after showing protective antiviral activity against SARS-CoV-2 and MERS-CoV, among others, in laboratory experiments.
“These proteins are active against all coronaviruses,” O’Keefe said.
Searching for Pandemic Solutions
O’Keefe explained that, back in 2010, when SARS-CoV-1 was circulating, MTP published findings showing that griffithsin could protect animals against infection by this virus, as well as several other human coronaviruses.
In 2020, the griffithsin paper started to gain fresh attention from virus researchers, and O’Keefe, who suspected that CV-N worked in a similar fashion as griffithsin against enveloped viruses, heard from his longtime collaborators who wanted to see whether they could work together to find some solutions for the COVID-19 pandemic. They decided to see whether CV-N might work against SARS-CoV-2.
MTP brought to the table its expertise in studying protein and small-molecule interactions. The groups in Brazil and Spain brought their experience in producing large amounts of active CV-N using plant-based systems, their testing facilities, patient virus samples, specialized assays, and the means to assess the effectiveness of CV-N drops in specific testing models. “It was a very international collaboration,” noted O’Keefe.
The results of their collective efforts are cause for optimism, though more work remains to be done. One problem is that CV-N can be toxic to cells under certain circumstances. However, Krumpe pointed out that lectins can be tweaked to avoid this. Another limitation is that the group’s study occurred only in cell samples and animal models, not human patients.
Still, if the findings translate to humans, different lectins have different interactions with viruses—and those differences can be potentially exploited if different variants of concern emerge, Krumpe said.
For instance, CV-N had stronger antiviral effects on the SARS-CoV-2 Delta and Omicron strains than the earlier strains. As the virus evolved to become more transmissible in humans, it became more vulnerable to CV-N.
Also, earlier research on the use of CV-N against HIV showed that enough CV-N could be inexpensively produced in plant-based systems, such as soybeans, rice, and tobacco, to support clinical trials. Producing large quantities is a viable option and would be especially helpful for pandemic preparedness in developing nations.
Despite the work that will be needed to make CV-N an option for human use, O'Keefe is optimistic about the utility of developing it, and other antiviral lectins, to use as tools to manage future outbreaks, stating that the hope is “if any new coronaviruses crop up 20 years down the road, these [lectins] would still likely be active against them and be able to help prevent the early stages of a pandemic—the spread of the virus.”
Video highlights of these findings, produced by EMBRAPA/National Institute of Science and Technology Synthetic Biology, Brazil, are available via this link: Cyanovirin-N: A lectin that can help block the spread of COVID-19.
Lisa Simpson is a technical editor in Scientific Publications, Graphics & Media (SPGM), where she edits corporate reports, client projects, and scientific manuscripts, and writes for the Poster newsletter. SPGM is the creative services department and hub for editing, illustration, graphic design, formatting and multimedia training and support for NCI Frederick and Frederick National Laboratory.