By Karen Surabian, Thomas Stackhouse, and Jeffrey W. Thomas, Contributing Writers
As the fall and winter seasons progress, you may be attending more scientific conferences, where you may find a number of opportunities for research collaborations.
To assist your lab in reaching its research goals through collaborations, the staff of the National Cancer Institute Technology Transfer Center (NCI TTC) can guide you through a tool box of agreements you may need for protecting your intellectual property (IP) and effectively managing your collaboration.
NCI TTC is the IP and partnership liaison for the NCI laboratories and scientists. NCI TTC manages the NCI patent portfolio and negotiates transactional agreements with outside parties (universities, pharmaceutical and biotechnology companies, and other research organizations) to formalize collaborative partnerships.
Employee Discovery and Invention Reports (EIRs) describe your invention, list the possible inventors, and assign patent ownership to the U.S. government.
Managing discoveries through future research collaboration must be balanced with any potential commercial development. Moving your discovery into the hands of physicians and patients, where it can be of most benefit, is a critical, yet complex, process requiring a team of experts from many different fields. The starting line for this process is the reporting of your discovery.
A reportable invention includes, but is not limited to, organisms, such as mice, as well as compounds, assays, machines, software, methods of manufacture, and methods of use.
A disclosure may include, but is not limited to, posters, presentations, abstracts and journal articles (including electronic), and even job interviews with non-NIH personnel. You are advised to submit an EIR before—preferably three months before—a disclosure.
To ensure that the EIR is filled out and managed properly, your NCI TTC specialist will assist you in completing the EIR form; coordinate the reviews of EIRs; work on your behalf with the NIH Office of Technology Transfer regarding filing and licensing of your patent applications; and work with other NIH offices such as the Office of General Counsel and other NIH institute technology transfer offices.
Types of Collaborative Agreements
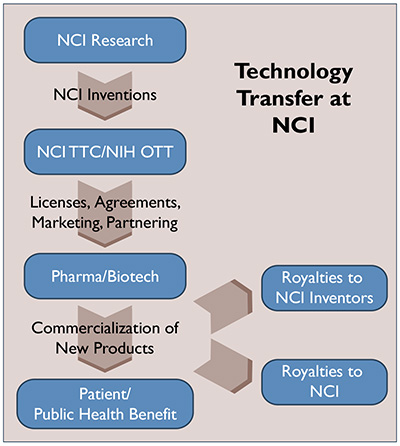 NCI TTC staff is experienced in negotiating several types of transactional agreements, as outlined below. Your situation will be assessed individually, so that the proper agreement(s) may be drafted with your goals, as well as those of your collaborator, in mind.
NCI TTC staff is experienced in negotiating several types of transactional agreements, as outlined below. Your situation will be assessed individually, so that the proper agreement(s) may be drafted with your goals, as well as those of your collaborator, in mind.
1. Confidential Disclosure Agreements (CDAs) are used for various situations in which confidential, proprietary, and/or unpublished information will be disclosed between parties. The flow of the information may be in one direction (e.g., from your laboratory to the collaborating organization) or may be in multiple directions (e.g., back and forth between your laboratory and multiple collaborating organizations).
Disclosure of confidential information before filing a patent application may have an adverse impact on certain patent rights and other IP issues. This is especially true now that we are operating under the new patent law of the America Invents Act. Therefore, when submitting your EIR, you also should think about your communications associated with unpublished information/data. That is where the Confidential Disclosure Agreement (CDA) becomes important to protect your work.
If you are considering disclosure of confidential proprietary and/or unpublished information to an outside party, please contact NCI TTC for assistance in executing the most appropriate agreement.
2. Material Transfer Agreements (MTAs) are used to exchange proprietary research materials either into or out of NCI and to document the source of the materials. These agreements are important because they offer NCI scientists protection related to issues of publication and use of materials for research purposes; MTAs may also include terms to protect confidential and/or unpublished information.
NCI TTC recognizes that the exchange of important, and often unique, research materials is critical to your work. Some materials require special attention because they may be related to human subject materials or other special circumstances. Before transferring or exchanging materials with outside organizations, contact NCI TTC, whose staff is trained to navigate you through any unique transfer situations.
One special type of MTA is a Collaboration Agreement (CA), which contains a jointly defined research plan and permits the exchange of confidential information, as well as materials, both to and from the collaborator.
3. Clinical Trial Agreements (CTAs) are used for studies conducted in humans to determine the safety and efficacy of agents and devices brought in from outside parties.
If your laboratory becomes involved in working on research that leads to clinical trials, you should contact NCI TTC for help in negotiating CTAs. These agreements enable you to transfer a patented, investigational agent only from a collaborator to NCI for NCI-sponsored clinical trials.
CTAs offer no promises to future IP rights, but do allow for access to data for filings with the U.S. Food and Drug Administration. These agreements are executed with the expectation that the collaborator has already completed preclinical and Investigational New Drug–directed toxicity studies. They specify the use of the investigational agent in humans and reporting requirements for adverse drug reactions.
4. Cooperative Research and Development Agreements (CRADAs) are collaboration agreements, usually with a commercial partner, that specify terms of confidentiality and an exclusive option to the collaborator to license future inventions. The CRADA allows for funds to be provided to the NIH laboratory to support its part of the research being conducted under the CRADA. However, NIH is not permitted to provide funding to the outside collaborator under the CRADA.
CRADAs provide for the exchange of material, equipment, and personnel, and are used when NIH and industry collaborate to develop a technology for commercialization. There are many types of CRADA collaborations, and NCI TTC will assist your team in navigating them and negotiating the best terms to protect your research while advancing the discovery.
If You Are Not Sure, Ask
If you have any questions or are not sure which agreement is appropriate for your research situation, contact the NCI Technology Transfer Center office:
Telephone: 301-845-5465
Website: http://ttc.nci.nih.gov
Follow Us on Twitter
http://twitter.com/NCITechTransfer
Join our LinkedIn Group
http://www.linkedin.com/groups/NCITechTransfer-NCI-Technology-Transfer-Network-4688463/about


