Platinum Highlight Abstract - November 2013
Characterization and Favorable In Vivo Properties of Heterodimeric Soluble IL-15?IL-15R? Cytokine Compared to IL-15 Monomer
Elena Chertova, Cristina Bergamaschi, Oleg Chertov, Raymond Sowder, Jenifer Bear, James D. Roser, Rachel K. Beach,
Jeffrey D. Lifson, Barbara K. Felber, and George N. Pavlakis
J Biol Chem 288(25):18093-18103, 2013
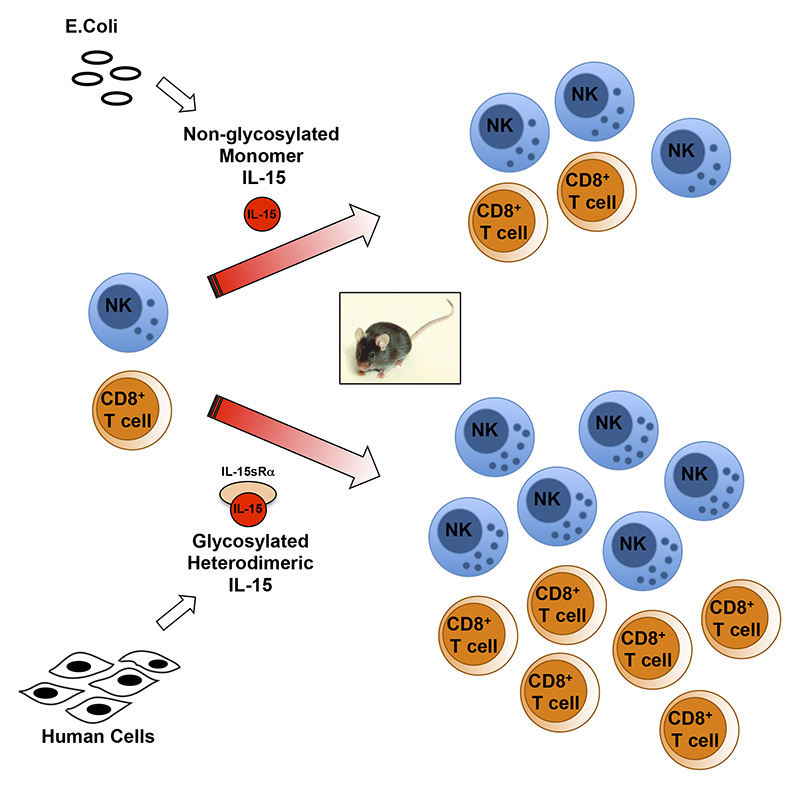
Interleukin-15 (IL-15), a 114-amino acid cytokine related to IL-2, regulates immune homeostasis and the fate of many lymphocyte subsets. We reported that, in the blood of mice and humans, IL-15 is present as a heterodimer associated with soluble IL-15 receptor ? (sIL-15R?). Here, we show efficient production of this noncovalently linked but stable heterodimer in clonal human HEK293 cells and release of the processed IL-15•sIL-15R? heterodimer in the medium. Purification of the IL-15 and sIL-15R? polypeptides allowed identification of the proteolytic cleavage site of IL-15R? and characterization of multiple glycosylation sites. Administration of the IL-15•sIL-15R? heterodimer reconstituted from purified subunits resulted in sustained plasma IL-15 levels and in robust expansion of NK and T cells in mice, demonstrating pharmacokinetics and in vivo bioactivity superior to single-chain IL-15. These identified properties of heterodimeric IL-15 provide a strong rationale for the evaluation of this molecule for clinical applications.

